- 1. Introduction to neuroethics
- 2. Introduction to neurogenetics
- 3. Diagnostic uses of neurogenetics
- 4. Genetic testing
- 5. Behavioral uses of neurogenetics
- 6. Genetic enhancement
- 7. Are we practicing eugenics?
- 8. Medical vs. social definitions of problems and solutions
- 9. Some final considerations
- 10. Neurogenetics in popular culture and in the popular imagination
- 11. Case studies
- 12. Conclusion
- 13. References
2. Introduction to Neurogenetics
2.1. Definition
Neurogenetics is a branch of genetics that analyzes the impact of genes on the structure and function of the brain and peripheral nervous system. Because studies of neurogenetics pertain to genes that determine our individuality, as well as diseases and dysfunctions, exploration of normal and disordered genes in the nervous system requires further knowledge and thoughtful consideration.
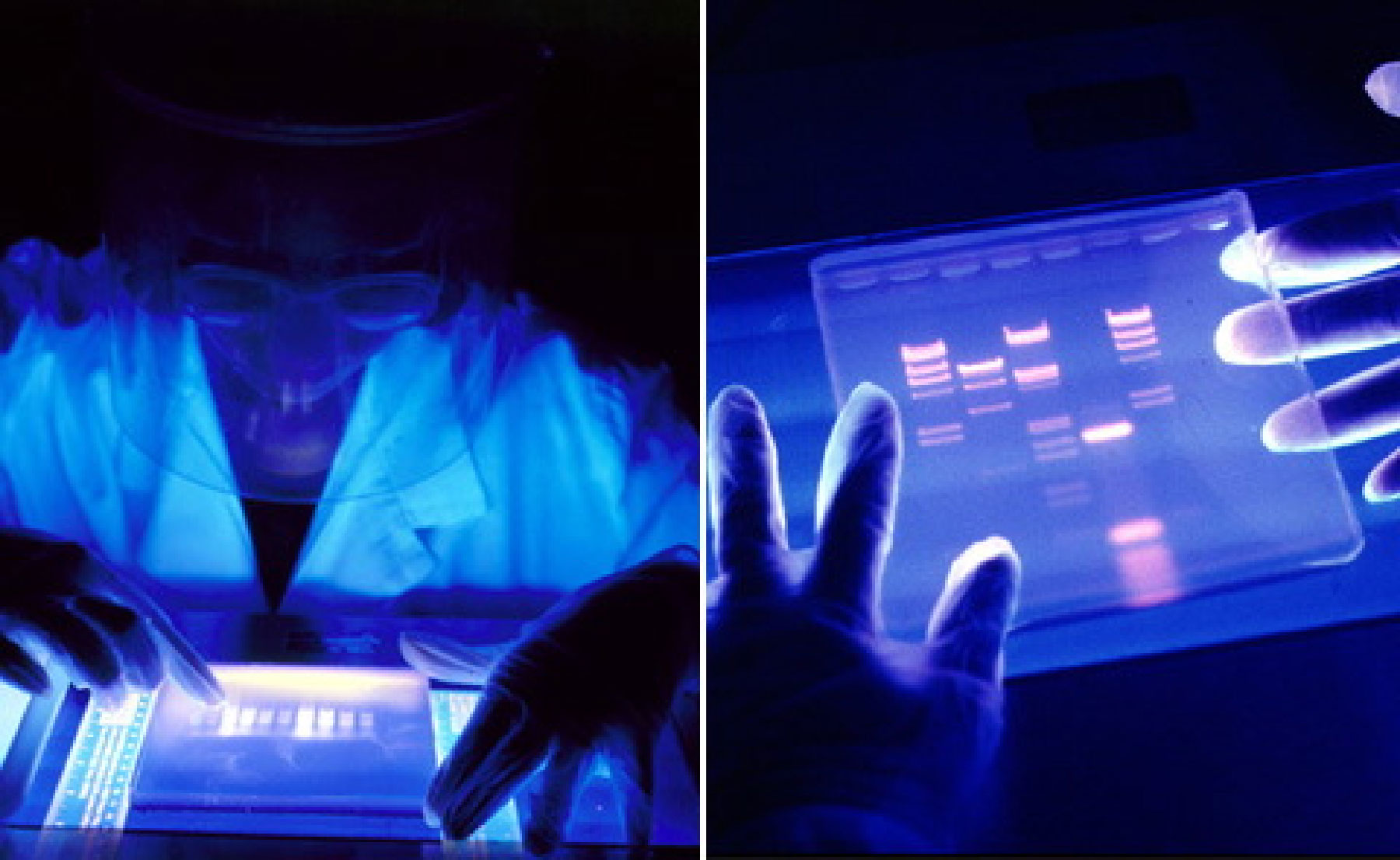
Source: Australian Genome Alliance
One aspect of neurogenetics identifies the genetic basis for diseases and pathologies of the brain and nervous system. This information could be used to identify factors that cause or increase risk for those conditions. Subsequent steps could be taken to prevent or mitigate the onset of symptoms, or at least enable genetic counseling that could protect the gene-bearer's offspring from the condition.
A second aspect of neurogenetics identifies the genes associated with various positive personality traits and abilities (musical talent, intellect, athletic ability), as well as negative traits such as violence and aggression.
2.2. Reasons for ethical consideration
All new technologies deserve reasoned, thoughtful debate prior to large-scale introduction, whether the technologies or the debates are biomedical in nature, legal, political, even environmental. If an innovation has the potential to change the ways in which we live and work, it is essential that we consider the ethical, social, and legal implications and decide if, on balance, the positive outcomes outweigh the negative.
Are the potential benefits worth the potential costs, efforts, or harms?
3. Diagnostic uses of neurogenetics
3.1. Finding disease markers
The National Institute of Neurological Disorders and Stroke (NINDS) estimates that twenty percent of the United States population, about 50 million people, suffer from at least one of more than 600 neurological diseases.2 Many of these disorders have been linked to genetic factors, while for others the etiologies and whether or not they are genetically determined are still unknown. The same points can be made with regard to psychiatric disorders which are among the most prevalent disorders world wide. Indeed, advances in genetics have brought the fields of neurology and psychiatry closer together than ever before.

There is considerable interest in the role of genetics in neurological diseases. In some cases, we only know that a correlation exists between a region on a chromosome and a disease. In other cases, specific mutations have been identified that cause the disease in question (e.g., Huntington's disease, Fragile X syndrome). In many instances, mutations have been identified that increase the risk for a disease even though they have not been proven to be a direct cause of the disease (e.g., APOE4 in Alzheimer 's disease). Even in conditions where we know of specific genetic mutations (e.g., in Parkinson's disease, amyotrophic lateral sclerosis (ALS), muscular dystrophy, and many forms of epilepsy), it has become clear in recent years that the genetics of many of these common disorders is complex in that many genes contribute to the disease phenotype.
It is important to emphasize that we are dealing with brain disorders. As we learn more about genetic determinants, traditional distinctions between neurological and psychiatric diseases may fade or disappear. Genes may determine traits in addition to defined clinical disorders. The depression of Parkinson's disease may have the same determinants as the depression observed in bipolar disorder.
In this module, we present a brief overview of the genetics of some common neurological disorders and the ethical implications for the individual and society that this fast moving field holds. The aim is to encourage thoughtful debate about the positive as well as negative uses and implications of neurogenetics.
3.1.1. Huntington's Disease
Through the field of neurogenetics, we can test for genetic markers and mutations for pathologies in the brain. The classic example is Huntington's disease (HD), a neurodegenerative brain disorder that is both monogenetic and has complete penetrance. At the present time, it is inevitable that those who carry the HD mutation will develop this disease if they live long enough, and a parent who carries the HD mutation (excessive CAG repeats) has a 50:50 chance of passing the HD mutation to each of his or her children.
A genetic test for Huntington's disease (HD) is currently available to at-risk persons with affected family members. Because the test is for a single gene mutation with high penetrance, those who test positive for the HD mutation will inevitably exhibit degeneration of nerve cells in the basal ganglia. People at risk for HD may choose to be tested, but many choose not to be tested. Even among those who do undergo testing for the HD mutation, many then choose not to learn their results if they feel they cannot face the burden of knowing their genetic status.
3.1.2. Autism
Autism provides a stark contrast to Huntington's disease. It is clearly a genetic disorder in many families, but no single gene has emerged that can portend this devastating developmental disorder. The emphasis on genetics comes from the simple fact that the concordance rate for autism among identical twins is extraordinarily high--between 70% and 90% in different studies. The complexity of the genetics is indicated by the fact that the phenotypes of concordant identical twins may vary widely--from mildly to severely affected.3
Current efforts are focused on early diagnosis, even though it is difficult to be certain of an autism diagnosis before the age of 3, as verbal IQ and mild language delays may complicate routine clinical evaluations. In working with toddlers, evaluators use measures that are independent of verbal interaction such as tracking eye movements and on estimates of brain volume obtained by non-invasive Magnetic Resonance Imaging (MRI). Attention is focused on infants (6-12 months of age) who have older siblings with a clear diagnosis of autism. The likelihood of developing autism is higher in such "infant siblings" than in the general population. The importance of early diagnosis lies in the fact that prognosis may improve with early intervention.
Although a genetic test might enable early diagnosis of autism, genome scans of families (linkage analyses) or populations of unrelated individuals (association analyses) have been inconclusive. Suspicious regions have been identified on many of the 23 pairs of chromosomes, but few of these "hot spots" have been seen in more than one study.4 Thus, definitive, reproducible molecular targets have yet to emerge in autism. We do not yet have a reliable diagnostic, biochemical or molecular test for the majority of cases of autism.
One reason for the lack of definitive genetic findings lies in the complexity of the clinical phenotype. The DSM IV and other diagnostic manuals now place classic autism among a larger class of Progressive Developmental Disorders. There may be several forms of autism. Classic autism is characterized by deficits in three principle areas: social cognition, language development, and repetitive, restrictive movements. Each of these categories shows wide variation. Social cognition deficits, for example, can vary from infrequent eye contact to total lack of interpersonal play and empathy, as well as total inability to engage in tasks that require joint attention.
Another reason for the lack of genetic answers may lie in the complexity of the human genome. In recent years, we have learned that there is variation in the numbers of many different genes. We presumed that individuals have two copies of each gene, one on each chromosome, but recent data have uncovered "copy number variants" (CNVs). Small (sub-microscopic) regions of a chromosome may be duplicated one or more times, and others may be deleted leaving one copy, or none at all. Genetic mutations may be due to CNVs as well as to "mis-spellings" or changes in the nucleotide sequence. Techniques for detecting CNVs have evolved rapidly, and we can expect detection tests to become more routine in the near future.
Children diagnosed with Fragile X syndrome or Rett Syndrome often exhibit features of autism. It is therefore recommended that children diagnosed with autism also be tested for the Fragile X and Rett syndrome mutations. The presence or absence of a genetic mutation may affect the types of care and treatment an individual receives. Most clinicians agree that intervention with behavioral therapy for children with autism is most effective when started early; therefore, if doctors learn of the presence of a Fragile X mutation, they may begin more tailored interventions. Other co-morbidities have been identified, and they may be added to the list of recommended tests in the future. Of interest, such "syndromic" autism may be associated with single gene defects that may shed light on other cases of "essential" (no co-morbidity) autism.
3.1.3. Alzheimer's disease
Genes play key roles in Alzheimer's disease (AD) but the roles are different for early onset AD and late onset AD. Early onset disease usually begins between age 45 and 60, and certainly before age 65 if there is a strong family history of the condition. Indeed, early onset AD is often referred to as Familial Alzheimer's disease (FAD). FAD is relatively rare, certainly much less common than late onset AD. It is inherited in an autosomal dominant fashion. That is, the disease will occur if one mutant gene is inherited from either parent. Mutations in the Aß amyloid gene have attracted a great deal of attention. A fragment of this transmembrane protein called AB42 accumulates in plaques (dense clumps of abnormal proteins) that form throughout the brain. Plaques in the hippocampus and in the frontal lobes are particularly devastating for functions related to learning and memory. Ordinarily a shorter fragment, AB40, is produced that does not accumulate in plaques. A mutation in the AB gene, discovered in several families, leads to altered cleavage of the AB protein and accumulation of the dangerous AB42 fragment. Other genes can alter the processing of the parent AB protein. Two enzymes, beta and gamma secretase, regulate the cleavage of AB, and alterations in these enzymes can produce the toxic 43 amino acid cleavage product. Mutations in these enzymes have, indeed, been implicated in FAD.
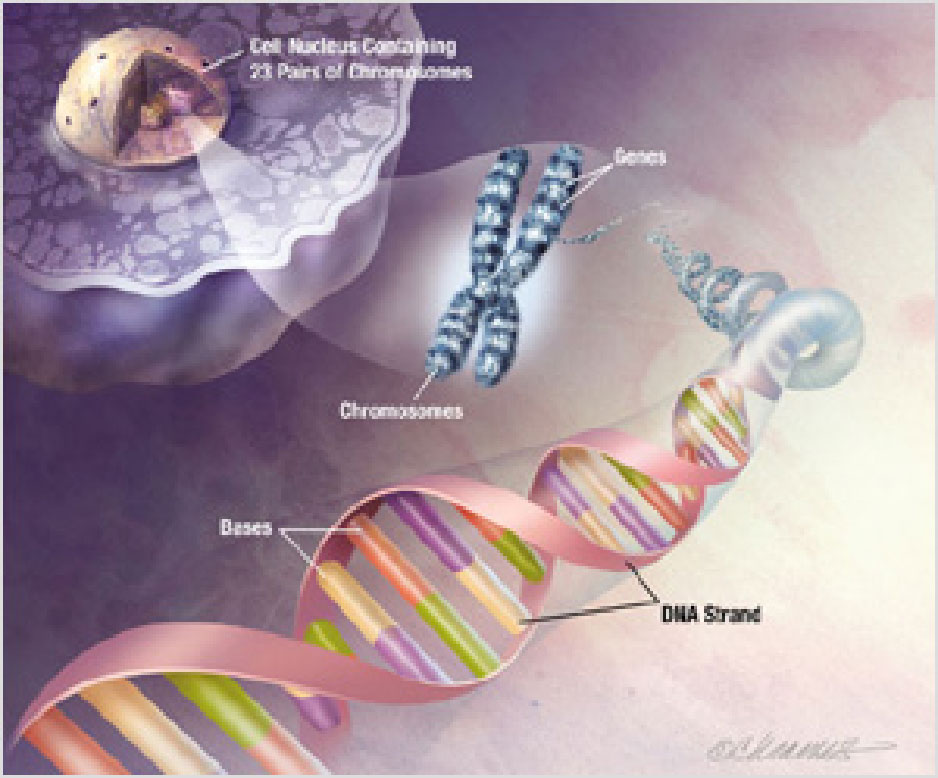
Source: National Institute on Aging
None of these genes has been implicated in the more common late onset form of the disease. The prevalence of late onset AD increases with each decade of life after age 65. Some estimates suggest that 50% of the population over 85 suffers from AD. Thus there is an urgent need to identify late onset AD risk factors. Despite the enthusiastic search for such identifiers, the complexity of the AD phenotype may be a confounding factor, just as the complexity of autism and other mental disorders is for genetic analyses of those conditions. We simply do not know how many biochemical mechanisms contribute to the various facets of the syndrome now called AD.
No genes have been identified that cause late onset AD. However, an allele of the APOE gene, located on chromosome 19, has been implicated as an important risk factor. Four alleles (#1 -- 4) of the APOE gene have been identified. They are all involved in the transport of cholesterol in the blood and in the metabolism of triglycerides. But something is different about the APOE4 protein. APOE4 is a risk factor, but it is not a certain predictor of the disease. Individuals who are homozygous for the E4 isoform may show no cognitive decline. Conversely, individuals with the E3 isoform which some consider to be protective for AD, may be severely demented.
APOE4 presents special problems for genetic counseling. It is not difficult to identify the various gene alleles of protein isoforms, but what does one advise a young couple who are both homozygous for the APOE4 isoform? As we have no effective treatment and no effective preventive measure that might delay the onset of AD, is the information useful? Should this be a consideration in their childbearing?
3.1.4. Epilepsy
Genes have been identified that are associated with several rare forms of epilepsy.5 However, the most common forms of epilepsy such as febrile seizures, post-traumatic epilepsy, and generalized absence seizures undoubtedly reflect the interplay of multiple genetic factors and the environment. Common forms of epilepsy do not follow a pattern of simple Mendelian inheritance. As with Alzheimer's disease, it appears that each risk conferring gene provides only a small additive effect to increase susceptibility.6
Many epilepsy genes encode components of membrane ion channels that regulate neuronal excitability. Indeed the different epilepsies are among a growing class of disorders now called "channelopathies." While such discoveries offer insights into underlying mechanisms, without more definitive causal links, there is limited utility to genetic testing of asymptomatic persons. This is the case even if the proband has a family history of the condition.
4. Genetic Testing
4.1. Genetic Testing of Minors
Many agree that presymptomatic, predictive testing of minors below the age of 18 for genetically-linked neurological conditions is not recommended for adult onset neurological disorders for which there is no prevention because the children and adolescents lack the capacity for truly informed consent. Since many adults with family histories that put them at risk for genetic-linked diseases choose not to avail themselves of these tests, or they choose to have the genetic test and subsequently decide not to receive the test results, it seems inappropriate to test children before they, too, have the opportunity to choose for themselves whether they want to know their genetic status with all the benefits and harms of this information.7
While there may be consensus not to test asymptomatic individuals before they reach an appropriate age of consent, there are exceptions to this rule -- notably with some forms of epilepsy where knowledge of the child's genetic status can be important in planning medication and treatment protocols. Genetic testing of symptomatic children is appropriate to obtain a definitive diagnosis. Lesch-Nyhan Syndrome, for example, is a particularly violent self-destructive disorder that often requires restraints and constant observation; genetic testing may distinguish it from other disorders associated with mental retardation to optimize therapy and predict prognosis.
4.2. Pre-natal testing and pre-implantation genetic diagnosis (PGD)
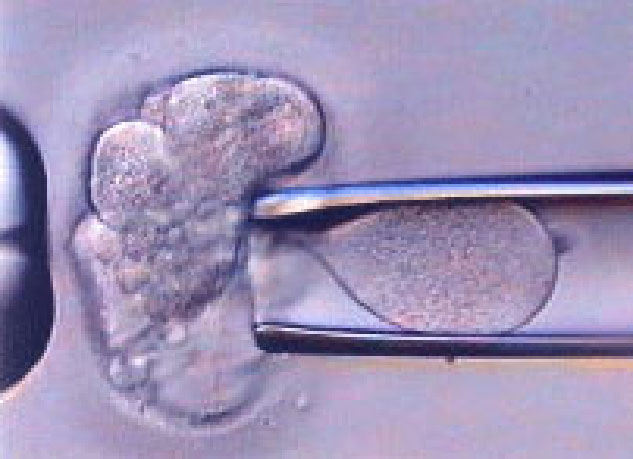
Source: International Institute on Gender Selection
Pregnant women and couples undergoing in vitro fertilization (IVF) procedures, especially those with a personal or family history of monogenic highly penetrant diseases, are often encouraged to undergo tests that reveal genetic information about the fetus or even the pre-implanted embryo. In cases where the tests show a genetic abnormality, but the parents decide to continue the pregnancy, what are the parents' responsibilities to the child in disclosing its genetic susceptibility to disease? Will they treat the child as if there were no genetic mutation, or rather, will they be overly cautious and overly protective with this child, a pattern that can lead to a Pygmalion effect, or even will they give up on the child prematurely?
4.3. Accuracy and predictability of results
Interpreting the results of genetic testing for many neurological diseases can be challenging, even for trained geneticists who are best prepared to interpret complex test results. Appropriate interpretation is challenged by varying knowledge of disease susceptibility, given certain genetic variants of unknown significance and gene interactions. One notable exception to this conundrum, of course, is Huntington's disease. To date, anyone having the grossly expanded CAG sequence will inevitably develop disease symptoms.
The likelihood of developing a neurological condition based on one's genetic profile or make-up is hardly a straightforward prediction. Researchers perform complex mapping of the many genes and many different alleles. A gene may indicate a 70% susceptibility for a trait or disease in one instance, but the presence of an accompanying gene may increase or decrease the likelihood of disease, offering an additive, a synergistic, or multiplicative effect. There are also complex environmental and psychological factors that can influence the likelihood of developing a disease or condition based on the genetic profile.
4.3.1. Perceived Risk

The imprecision of genetic screening for many neurological conditions and diseases is complicated by a fundamental misunderstanding of statistics on the part of the general population, or a refusal to accept statistical arguments -- even among the intellectually sophisticated. There is the concept of "perceived" risk as opposed to "actual" risk. As an example of this misunderstanding, take the well-established fact that car crashes result in more fatalities than airplane disasters. While the actual risk of riding in a car is much greater than in a plane, much of the public ignores this fact, and focuses more attention and anxiety on the perceived risks of air travel. Similarly, if a person has a gene that has a 20% chance of resulting in illness, will he or she emphasize that 20% chance of disease, or the 80% chance of health?
Studies have also shown that genes operate in complex networks rather than with simple, direct relationships. Instead of each sequence of DNA having a distinct and discrete function, it is now believed that genes and their effects interact and overlap.8 Predicting disease outcomes based on genetic profiles is therefore a complex matter, especially when one considers the environmental factors that contribute to disease etiology.
Source: Answers.com
Neurologists order many of the available genetic tests for neurological conditions. While knowledgeable and savvy about neurology, disease syndromes, and treatment modalities, the interpretation of genetic tests may be best done in partnership with trained geneticists and genetic counselors. Interpretation of many of the tests depends on a detailed knowledge of family history, on phenotypes associated with homozygous (both gene alleles are mutant) and heterozygous (one of the two gene alleles is mutant) states. Differentiating between benign normal genetic variants and disease-associated mutations can at times be difficult and may not be intuitively obvious to most neurologists. The manifestation of the same mutation may vary among individuals. It takes considerable experience to recognize all of the different forms that a given mutation might take. With enhanced understanding of the genetic data, the neurologist will be better positioned to work with the patients, provide additional testing when necessary, and to recommend and offer the most appropriate intervention or treatment.
4.3.2. Home Testing Kits
The availability of home testing kits allows individuals to screen themselves for certain genes and gauge their susceptibility for certain diseases. This kind of direct-to-consumer testing should be discouraged by physicians and geneticists. In addition to some tests not being valid, without proper interpretation or understanding of genetics and disease, a home test could result in needless confusion, panic, and erroneous contingency planning. Inaccurate or incorrectly interpreted home testing may also make people less vigilant about their other health maintenance issues (e.g., cardiovascular disease, diabetes, hypertension, etc.) that are more likely to cause morbidity or even mortality.

Source: HomeDNA.com
The availability of direct-to-consumer genetic testing kits has alarmed some physicians and genetic counselors. Just because something's available does not mean it's safe or effective or worth your money.
4.4. Informed Consent
Informed consent to conduct a specific genetic test should acknowledge "the potential social and family implications, including the potential for discrimination on the basis of genetic-risk status."9
When clinicians provide informed consent procedures for genetic testing for heritable neurological diseases, especially those for which there is no treatment or cure, they should consider the benefits and harms of the resulting information for the individual. In the case of a neurological disorder such as Huntington's disease, physicians should counsel patients on the utility of undergoing such a genetic test. For some, knowing that they will inevitably undergo a type of brain degeneration for which there is currently no cure, treatment, or effective prevention, may empower them to prepare for the future in terms of career choice, child bearing decisions, etc. For others, it would be too great a burden to bear this knowledge. For yet another subset of individuals, it can induce a sense of fatalism and futility in present and future endeavors, knowing one day they will become incapacitated.
4.4.1. Model requirements for informed consent
New York State law calls for eight statements that MUST be included in the informed consent process for patients undergoing clinical genetic testing. These eight required statements can serve as a model for an informed consent form. The statements include:
- A general description of the genetic test
- A statement of the purpose of the genetic test
- A description of diseases and conditions being tested for
- The likelihood of getting the condition given a positive genetic test result
- Recommendations for further clinical tests if the genetic test is positive
- A suggestion to seek genetic counseling prior to consenting to the test
- Individuals to whom the genetic test results may be disclosed
- Plans for destruction of the sample after a certain length of time so no other genetic tests may be done on the sample.
Additional suggestions for proper informed consent for genetic testing would include check-boxes that require research subjects/participants to actively select the uses they will allow for their tissue sample.10
4.5. Privacy and confidentiality
In recommending genetic tests for brain disorders, one must appreciate the implications and broad reaches of the resulting information and consider the possibility of a loss of privacy due to a breach of confidentiality in the cases of heritable diseases (as opposed to spontaneous genetic mutations). By recommending that an individual be tested for an inherited condition, you essentially suggest that a parent or a sibling may have the gene, and therefore this parent or sibling either may or will get the disease. Thus, these tests are only recommended for those who have a family history of or predisposition for contracting the disorder. There are also implications for the children of tested individuals, since a positive result for an inherited gene exposes the susceptibility of a child who may not want to know his or her genetic status or risk factors.
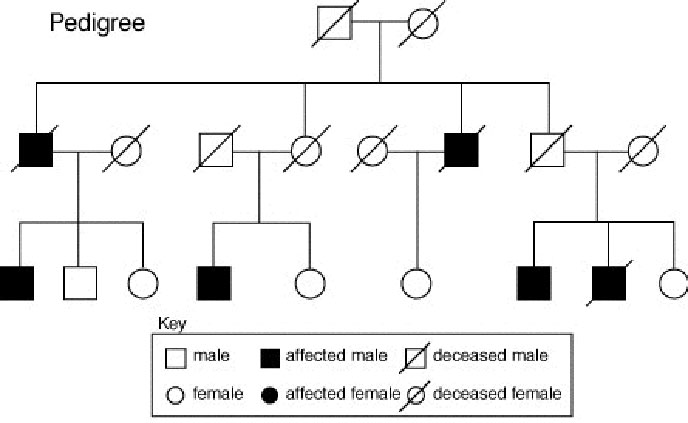
Source: Troy High School
Protecting privacy and maintaining confidentiality are critically important in neurogenetics. Information resulting from genetic tests reveals much about an individual and the individual's family. In addition, tests that reveal elements of a person's genetic code can, and often will, uncover unintended information with consequences that may affect more than just the individual. In about ten percent of a test group, truths about paternity are revealed that were not previously known. This information would be devastating information, indeed, were it released to inappropriate people (employers, insurance companies, etc.) through sloppy or careless privacy and confidentiality procedures.
The emergence of more easily-accessed genetic codes requires a full-scale revision of our current codes of privacy and confidentiality. There are questions about who, ultimately, owns the information displayed on an individual's genetic code. Is this information the property of the testing lab, the physician who ordered the test, the patient, the patient's family, etc.? Among these entities, whose responsibility is it to ensure that the information does not fall into the hands of insurers, employers, government agencies, and others who could use the information to discriminate against the individual? We must also consider the possibility of such information being used in the criminal justice system. The physician and the genetic counselor have a special obligation to protect the patient's right to privacy and to maintain strict confidentiality of private information.
Further, we must ensure that people do not undergo genetic testing without their knowledge. A patient's consent to a particular blood test is not an open-ended consent for every genetic test that can be done with a blood sample. For this reason, the informed consent process should require that the patient consent to specific tests or give permission for unspecified testing under certain circumstances, as well as the destruction and disposal of samples after a specified period of time.
4.5.1. Genetic exceptionalism
The notion of genetic exceptionalism recognizes the increased sensitivity of the results of genetic tests, as contrasted with other medical test results such as an X-ray or EKG. In the instance of neurogenetics, as with other genetic testing, the results of testing can contribute to negative labeling, and discrimination in insurance or employment.11 For this reason, many believe that the results of genetic testing hold a more privileged status with regards to privacy and confidentiality. The results of genetic tests can have far-reaching effects both for the individual and for first-degree relatives.

Source: Inno
Richard Ashcroft, in the British Medical Journal, discusses the need for genetic exceptionalism with regard to disclosure of genetic profiles to insurance companies,12 and further cites the possibility of discrimination based on actuarial misunderstanding of increased risk for certain diseases once a gene is identified. He also explains how actuarial computation may be scientifically sound but socially unjust. If the purpose of insurance is to protect the insured against ill luck, the insured should also be protected against the ill-luck of genetic predispositions. A counter argument presented by Soren Holm in the same issues suggests that if the individual seeking insurance knows that he or she is at a higher risk for a genetic disorder, then failure to disclose this information prevents the insurance industry from being able to make decisions that would create the most favorable risk-pool for the rest of their client population.13 Holm further points out that genetic information is already available and easily accessible through means such as family histories.
4.5.2. Certificate of confidentiality
When performing research of a sensitive nature, such as genetic testing, where the results could have adverse consequences for participants, researchers should apply for a Certificate of Confidentiality from the National Institutes of Health (NIH).
These certificates are designed to protect researchers, who may be served with a subpoena, from having to disclose identifying information about the research participants and test results for any civil, criminal, legislative, administrative, or other federal, state, or local court proceedings unless the participant consents. While these certificates have never been tested in court, it is assumed that the researcher cannot be compelled to reveal any information about the participants in his or her study. If the participant, on the other hand, reveals personal information to anyone, the certificate is no longer valid for that person.
As a further protection, many research organizations explain to their study participants in the informed consent process that the results of the genetic tests will not be released to the participants in the research study, usually because testing was not done by a CLIA approved lab and thus, if questioned in the future, the study participants are instructed to say that they have never undergone the genetic test.
5. Behavioral uses of neurogenetics
Source: Salisbury University
Research is currently exploring whether it is possible to identify genes for specific behaviors or characteristics e.g., intelligence, athletic ability, violence. It is important to recognize that genes with behavioral or personality implications would neither be monogenic, nor would they have complete penetrance. Despite the mathematically precise results demonstrated by Gregor Mendel in the 1800s, even within the realms and understandings of dominant and recessive genes, characteristics would not be predictably expressed. Additionally, there is a large human factor to consider in personality and behavioral traits, which brings up the familiar debate of nature vs. nurture. To some extent, people have the ability to mold their character traits, sometimes in spite of genetic predispositions.
5.1. Genetic Determinism
Persons who know their genetic make-up -- whether this make-up results from natural variation or is genetically planned at conception -- may allow greater expression of identified traits. A parent, knowing a child has the genotype (genetic propensity) for a trait may act on the expectation of that trait being exhibited, creating a self-fulfilling prophesy that the trait will be expressed much like the Pygmalion response.14 Those who believe in genetic determinism expect that characteristics manifest in their genetic profiles will be their destiny. This sense of inevitability ignores the very significant role played by environmental factors in shaping individuals and their personalities.
Behavioral choices and decision-making are complex processes of which genetics is only one element.15 Part of being human is overcoming impulses and behavior traits, and making independent intellectual decisions based on rational evaluations of the situation at hand. Revealing one's neurogenetic makeup can have deleterious effects by undermining personal enhancement and self-determination in favor of saying, "This is just how I was meant to be." A tragic exception is the Lesch-Nyhan Syndrome in which the individual (usually a child) is unable to control impulses to self-mutilate.16,17
6. Genetic enhancement
For the purposes of this section, enhancement can be defined as using technology to fundamentally change who we are for a socially-defined "better" state. This could include pharmacological preparations to make us sharper or smarter, or to enable us to require less sleep. It could include implants to improve otherwise normal hearing or vision. Relevant to neurogenetics, it could also include the alteration or selection of specific genes to make normal brains better. According to the National Human Genome Research Institute, this could one day be accomplished using injectable forms of the genes.18
Genetic enhancement experiments through the injection of specific genes or DNA sequences either into an adult organism or into an embryo have had limited success to date. This is due to the current lack of control once the genetic material enters the donor's cell nuclei and integrates into the host's DNA. There is usually little control of the number of gene copies inserted, or the specific site of integration of the donor DNA. Although these technologies may not be available for human experimentation and use for a long time, even at this early stage we should contemplate their meaning and future potential implications for society.
6.1. Genobility
There are concerns that genetic enhancement will likely be available only to the privileged classes. We must be wary of creating a "genobility," a class of people privileged precisely because of its genetic code.19 A type of natural genobility already exists to a certain extent: individuals choose partners and mates based on physical, intellectual, and behavioral attributes, and these traits are often class-related. The upper classes pass on their genes -- along with their social privileges -- in the form of opportunity, access to better health care and education, as well as access to life's better rewards. A truer genobility, created from deliberate rather than random DNA sequences, could preclude people from lower classes from ascending the social or economic ladder, despite personal dedication and successes.
7. Are we practicing eugenics?
While genetic enhancement refers to the injection or selection of genes for the purpose of improving the likelihood of good attributes, there are applications for genetic research and technology to remove negative attributes from society. Eugenics, literally meaning "well born," is a genetic and social theory whereby the human race is improved by selective reproduction in which desirable characteristics are propagated and undesirable traits are eliminated.
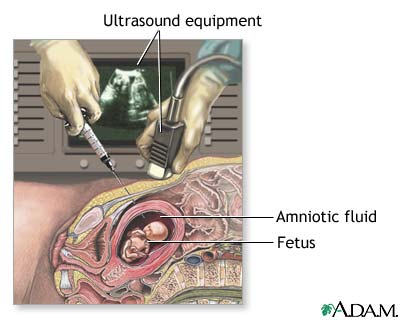
Source: Medline Plus
As an example, parents who carry genes related to disorders with high levels of penetrance may choose to undergo pre-implantation genetic diagnosis (PGD) to select an embryo without these genes so the resulting child would be unlikely to develop the disorder. Similarly, prospective parents may use amniocentesis to determine the fetus' genotype and choose not to continue a pregnancy if there is a high probability of a neurological disorder.
The use of these technologies to reduce the likelihood of a person being born with a severe neurological deficiency is relatively uncontroversial (e.g., Tay Sachs). The controversy arises when we decide what kinds of disorders or deficiencies are considered "severe." Many members of certain "disease" or disability communities may disapprove of parents who choose to use genetic screening technologies to eliminate their diseases or disorders in future generations (e.g., deafness, achondroplasia). These advocates assert that this choice de-values and de-legitimizes their own lives and the lives of their children who are born either with these disorders or with the increased possibility for developing them.
8. Medical vs. social definitions of problems and solutions
Neurogenetics may result in the medicalization or pathologization of natural human variation.20 Many traits that, in the extreme, are considered pathological can be detected in milder forms in the "normal" population, and there may not be a distinct dividing line between the two. Once science offers people the ability to control whether we have or we express a given gene, those who choose not to suppress it may be seen as additionally flawed. This could reinforce social prejudices and stereotypes to the detriment of people already in those stigmatized groups, as well as those for whom neurogenetic manipulation was not an option.
8.1. Designer Babies

Source: Savebabies.org
Questions of socially versus medically defined problems and deficiencies are important to consider in the growing world of pre-implantation genetic diagnosis (PGD). As described in Section 6 on Genetic Enhancement, PGD technology is currently used to:
- find euploid embryos for implantation, or
- screen out known fatal or serious disorders and deficiencies for which one or both parents are carriers or the couple's earlier child(ren) has(have) been diagnosed to have.
In the future, the ability to identify many more genes for different neurological, psychiatric, and behavioral traits and disorders before uterine implantation may present opportunities for parents to "design" their children, selecting in or out the desired physical, intellectual, and temperament traits they desire.
What is not certain, though, is if selecting the child's genetic make-up based on parental preferences is the right decision. It is possible that the world into which these designed children grow changes its values, leaving these genetically-designed children at a new disadvantage. Parents who have a socially undesirable trait, like deafness, may choose to reify their own existence by not sparing their children this trait, or even selecting for it, but the children may grow to resent their parents for it, wanting to be more like the norm.21 Other children may be "designed" to be genetically similar at the HLA locus to a sibling with a condition that can be "cured" with a bone marrow transplant. Once people "special-order" their "designer children," they will have different expectations of them. No longer will children be prized for their unique contributions to the world. In some cases, they may prove to be grave disappointments to their parents when they turn out to be different from what their parents "ordered" from their genetic menu. We also should remain cognizant of the differences between genotype and phenotype, remembering that just because someone has a gene for a particular trait, it does not mean that the trait will be expressed.
9. Some Final Considerations
The multiple uses of neurogenetics, both current and future, make the field an exciting one to study. Whether we use the findings to alleviate human suffering from neurodegenerative diseases, or use these findings to enhance the status of individuals and future generations, we can expect significant innovation in this area in the years to come. This module aims to encourage thoughtful debate about the positive as well as negative uses and implications of neurogenetics, with the goal of minimizing any of the harms while maximizing all of the benefits.
10. Neurogenetics in popular culture and in the popular imagination
Popular culture frequently jumps to the wildest extremes when depicting potential uses of emerging technology. Below are examples from "pop-culture" that stimulate popular imagination in neurogenetics. When considering these and others, we need to question if "the fantasy technologies . . . serve to dull the public's ability to be critical and thoughtful of actual new developments."22
10.1. The Boys from Brazil
The Boys from Brazil, by Ira Levin, is an example of how people conceive of genetic determinism and what it means. In the book, as the Nazis are losing the war, party officials decide to clone Adolph Hitler. The assumption is that it was Hitler's genetic make-up that made him, in their minds, the greatest ruler and visionary, and that by cloning him, they could create another great leader who could learn from the mistakes of the current regime and successfully carry out the mission of the Nazi party. The collaborators recognized that they needed more than just Hitler's genetic code to produce a new fuehrer, so they created several clones and set up each boy in a home situation mimicking the one in which Hitler was raised. When the Nazi hunters obtained the list of these boys, their ethical quandary was whether to take action against these still innocent pre-adolescents, or recognize that human and social factors can trump genetic codes in determining how people think and behave. In the end, they decided that destroying these boys on the basis of their genes and DNA would be akin to what the Nazis had done to six million Jews, so they allowed the boys to live their lives, rather than condemning them in advance for their genetic make-ups and for crimes they likely would never commit.

10.2. Gattaca
In the 1997 sci-fi movie thriller Gattaca, prospective parents are able to pre-select traits for their children. One couple decides to go the old-fashioned, natural, surprise route for their first son, at whose birth the doctors spew out the probabilities of his contracting a myriad of diseases and conditions that could have been prevented easily. Because of his genetics, and his related physical limitations, he is discriminated against in all aspects of his life, forcing him to assume someone else's identity in order to have more chances in life. He scrupulously avoids leaving behind any of his own DNA, requiring extreme exfoliation, keeping his hair very short, etc. Under this assumed identity and using his alias's blood, urine, and skin in his daily genetic ID tests, he is able to succeed in an aerospace career that would have been closed to him based on his own genetic make-up.

Please click here to go to the interesting cases and challenging questions.
1 Dinges D. Cited in Moreno JD. Juicing the Brain: Research to limit mental fatigue
among soldiers may foster controversial ways to enhance any person's brain. Scientific
American. 2006. November 29.

2 National Institute of Neurological Disorders and Stroke [Web site on the Internet]. Brain
Basics: Know Your Brain. [cited 2007 Aug 8]. Available from:
http://www.ninds.nih.gov/disorders/brain_basics/know_your_brain.htm.

3 Ma DQ, Cuccaro ML, Jaworski JM, Haynes CS, Stephan DA, Parod J, Abramson RK,
Wright HH, Gilbert JR, Haines JL, Pericak-Vance MA. Dissecting the locus heterogeneity
of autism: significant linkage to chromosome 12q14. Molecular Psychiatry. 2007 Apr;
12(4): 376-84.

4 Eliez S. Autism in children with 22q11.2 deletion syndrome. Journal of the American
Academy of Child & Adolescent Psychiatry. 2007; 46(4): 433-4.

5 Barral-Rodriguez S. Genetic Mapping of Complex traits: Efforts in Schizophrenia and
Alzheimer's. Presentation, Columbia University Seminar in Genetic Epidemiology. 2007
June 14.

6 Shostak S, Ottman R. Ethical, legal, and social dimensions of epilepsy genetics. Epilepsia. 2006 Oct; 47(10): 1595-602.
7 Ottman R. Analysis of genetically complex epilepsies. Epilepsia. 2005; 46(suppl.10): 7-
14.

8 Craufurd D, Donnai D, Kerzin-Storrar L, Osborn M. Testing for children for `adult' genetic
diseases. Lancet. 1990 June 9; 335: 1406.

9 Caruso D. A Challenge to Gene Theory. A Tougher Look at Biotech. The New York
Times. 2007 July 1. Available from:
http://www.nytimes.com/2007/07/01/business/yourmoney/01frame.html?ex=1188619200
&en=2c569f0a9d9aefbb&ei=5070.

10 Burke W. Genetic testing. N Engl J Med. 2002 Dec 5; 347(23): 1867-75.

11 Public Responsibility in Medicine and Research (PRIM&R). Wrap-up of Webinar: Protocol
Review in the New Era of Genetics. PRIM&R Membership Newsletter. 2007 July 31.

12 Shostak S, Ottman R. Ethical, legal, and social dimensions of epilepsy genetics.
Epilepsia. 2006 Oct; 47(10): 1595-602.

13 Ashcroft R. Should genetic information be disclosed to insurers? No. BMJ. 2007 June 9;
334: 1197.

14 Holm S. Should genetic information be disclosed to insurers? Yes. BMJ. 2007 June 9;
334: 1196.

15 Green RM. From genome to brainome: charting the lessons learned. Neuroethics:
Defining the Issues in Theory, Practice, and Policy (J Illes, ed.). New York: Oxford
University Press; 2006.

16 Churchland PS. Moral decision-making and the brain. Neuroethics: Defining the Issues in
Theory, Practice, and Policy (J Illes, ed.). New York: Oxford University Press; 2006.

17 Preston R. An Error in the Code. The New Yorker. 2007 August 13, p. 30.

18 Jinnah HA, Visser JE, Harris JC, Verdu A, Larovere L, Ceballos-Picot I, Gonzalez-Alegre
P, Neychev V, Torres RJ, Dulac O, Desguerre I, Schretlen DJ, Robey KL, Barabas G,
Bloem BR, Nyhan W, De Kremer R, Eddey GE, Puig JG, Reich SG. Delineation of the
motor disorder of Lesch-Nyhan disease. Brain. 2006 May; 129(Pt 5): 1201-17.

19 National Human Genome Research Institute [Web site on the Internet]. Genetic
Enhancement. [updated 2006 April; cited 2007 Sept 26]. Available from:
http://www.genome.gov/10004767.

20 Greely HT. Seeking More Goodly Creatures. Cerebrum. 2004; 6(4): 49-57. Avalable from:
http://www.dana.org/news/cerebrum/detail.aspx?id=1086.

21 Parens E. Creativity, gratitude, and the enhancement debate. Neuroethics: Defining the
Issues in Theory, Practice, and Policy (J Illes, ed.). New York: Oxford University Press;
2006.

22 Klitzman R. Clinicians, patients, and the brain. Neuroethics: Defining the Issues in
Theory, Practice, and Policy (J Illes, ed.). New York: Oxford University Press; 2006.

23 Grainger-Monsen M, Karetsky K. The mind in the movies: a neuroethical analysis of the
portrayal of the mind in popular media. Neuroethics: Defining the Issues in Theory,
Practice, and Policy (J Illes, ed.). New York: Oxford University Press; 2006.

24 Cuomo A. NY Hospitals Agree To Informed Consent About Genetic Testing. North
Country Gazette. 2007 April 3. Available from:
http://www.northcountrygazette.org/articles/2007/040307GeneticTesting.html.

25 Burke W. Genetic testing. N Engl J Med. 2002 Dec 5; 347(23): 1867-75.

26 Johnson A. Genetics and Health Insurance. Genetics Brief: Briefing Papers on Important
Genetic Issues of the Day. 2002 June. Available from:
http://www.ncsl.org/programs/health/genetics/Geneticshealthins.pdf.

References for Sidebars
S1 Kim J, Inoue K, Ishii J, Vanti WB, Voronov SV, Murchison E, Hannon G, Abeliovich A. A MicroRNA
Feedback Circuit in Midbrain Dopamine Neurons. Science. 2007 Aug 31;317(5842):1220-4.


